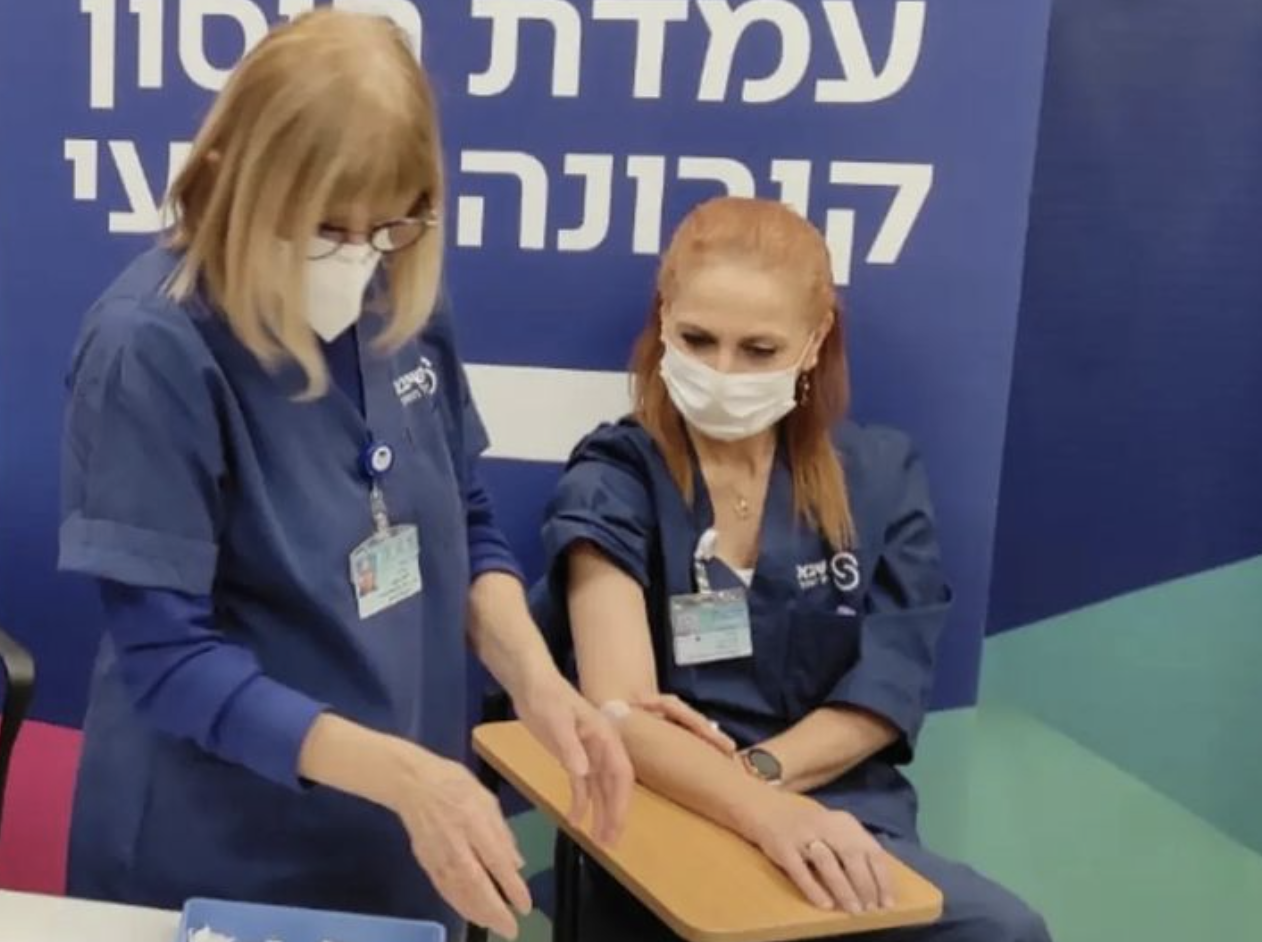
As of today, Monday, December 27, the clinical trial began with vaccinating the fourth dose of the vaccine Corona Virus. Israel is the leading country in the initiative. Specifically, 150 employees of the Sheba Medical Center in Ramat Gan, outside Tel Aviv, were selected for the tests. A hospital spokesperson explained that the study “will focus on the efficacy and safety of the vaccine in antibody production, in order to ascertain whether a second prolongation would be beneficial or not.” booster for the general population.” The opportunity to conduct a fourth dose has already been under discussion among national health authorities for a few weeks, to protect people at risk from the new wave of infection, fueled by the Omicron variant. Israel is the first country in the world to go forward with a double booster Vaccination, and among the categories that can participate as a priority, there are over 60, health professionals and individuals with severely compromised immune systems.
About 45% of the Israeli population has currently received the third dose of the vaccine. While 2,000 cases of Omicron have already been sequenced in the country, two of Sheba’s 150 hospital employees are vying to register the first Israeli to receive the fourth dose. On the hospital’s Facebook page, around lunchtime, a video of the nurse, Orna Rakhminov, was uploaded, as “the first woman to receive the fourth dose.” News agencies and newspapers responded to the news that this is the first Israeli country to receive double this amount booster. In fact, soon after, came the refusal posted by another employee of Sheba, Professor Jacob Lavie. for radio station may be, the man specified that Rakhminov “was the first woman,” but he was the first hospital employee to receive the fourth dose of the vaccine.
Read also:

“Freelance social media evangelist. Organizer. Certified student. Music maven.”